Featured as a back-of-page article in the CSABC Quarterly Letter of March 2015
by Richard Peachey
It was my second year in college, about twenty years ago. I was taking a science degree, and I had helped to arrange a creation/evolution debate on campus. Late in the evening one of the debaters injected a brief statement about right-handed amino acids having been found in some organisms. This stuck in my craw, because it seemed to strike a blow at a common creationist argument based on the “Chirality Problem” — thus weakening the case for creation.
What is the Chirality Problem? All proteins in our cells (and every organism’s cells) are composed of building blocks called amino acids. In general, an amino acid comes in two forms having all the same parts: bonded to the central carbon, C, are (1) an amino group, NH2; (2) a carboxyl (acid) group, COOH; (3) a hydrogen atom, H; and (4) the remainder of the molecule (“R” group), which is different for each type of amino acid.
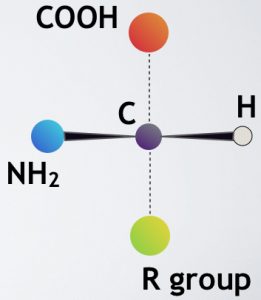
General structure of a left-handed amino acid
The two forms of an amino acid, although they have exactly the same parts, are built differently; they have opposite three-dimensional bonding arrangements. This is much like your left and right hands: they have all the same parts but are structured differently; in fact they are mirror images of each other. For this reason amino acids are called “chiral” molecules — meaning “handed,” from the Greek cheir, “hand.”
(Chemists call the two forms enantiomers — Greek for “opposite parts” — and describe them as non-superimposable, meaning that you can’t make one look exactly like the other by just turning it around. The amino acid glycine is an exception: it is achiral — not chiral — because its “R” group is simply a hydrogen atom.”)
The two versions of a given amino acid are energetically (or thermodynamically) equivalent — that is, they are equally easy to form from precursor molecules. They share many properties in common: they both have exactly the same density, melting point, boiling point, solubility, and spectroscopic properties. (The two forms of an amino acid do differ in two ways: they tend to interact differently with other chiral molecules; and they rotate plane-polarized light in opposite directions, as illustrated below.)
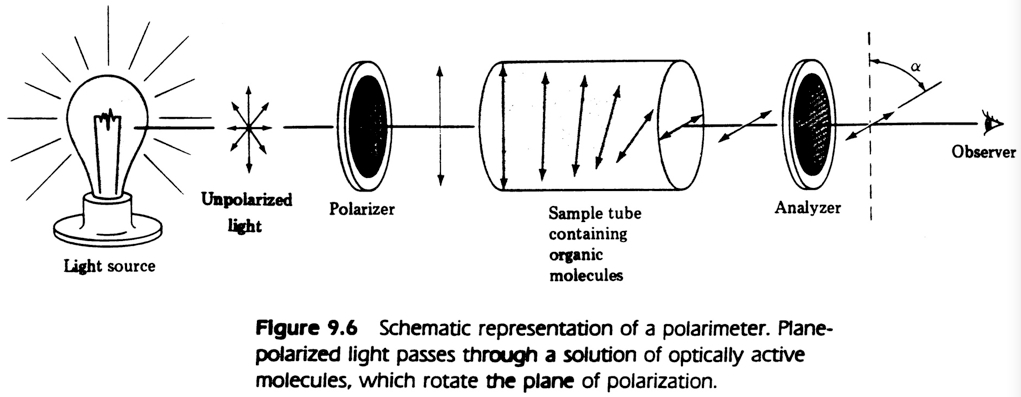
from John McMurry, Organic Chemistry, 3rd edition. Pacific Grove, CA: Brooks/Cole, 1992, p. 292
Because of this energetic equivalence, when amino acids are formed by natural chemical processes outside cells, the result is always a 50-50 mixture of right- and left-handed molecules (“racemic mixture”).
“. . . all known laboratory abiotic synthetic processes result in racemic mixtures of amino acids. . . .” (Science 279:363, Jan. 16, 1998).
“. . . when chiral molecules are synthesized in the laboratory from achiral building blocks, equal amounts of L- and D-enantiomers are produced unless painstaking care is taken to introduce an asymmetric agent during during the synthesis.” (Scientific American 262(1):110, Jan. 1990).
But proteins in living cells are always composed entirely of left-handed amino acids! There is no known explanation of why proteins should consist exclusively of left-handed amino acids if life originated through random unguided chemical interactions in a “primordial soup,” as hypothesized by evolutionary origin-of-life chemists.
“. . . there are no apparent biochemical reasons why L [left-handed] amino acids should be selected over D [right-handed] amino acids.” (Science 279:363, Jan. 16, 1998).
“The origin of homochirality, that is, the almost exclusive one-handedness of the chiral molecules found in terrestrial organisms, is a key problem in the origin of life.” (Science 275:951, Feb. 14, 1997).
“The origin of one-handedness in biological molecules is not yet clear. Several explanations have been put forward to explain how homochirality came about, but all are speculative — it is not even known yet whether it arose by chance or by some other means.” (Nature 409:777, Feb. 15, 2001).
“‘I spent 25 years looking for terrestrial mechanisms for homochirality and trying to experimentally investigate them and didn’t find any supporting evidence,’ said [organic chemist William] Bonner [professor emeritus at Stanford University]. ‘Terrestrial explanations are impotent or nonviable.'” (Science 267:1265, Mar. 3, 1995).
So this is the Chirality Problem, and it has greatly challenged evolutionists. But it’s also true, as the debater stated, that right-handed amino acids have been found in some organisms.
The most famous examples are the D- (right-handed) amino acids incorporated into various bacterial cell walls; they have also been found in bacterial capsules and microbial antibiotic products. Bacterial cell walls often incorporate short amino acid sequences (such as tetrapeptides, chains of four amino acids) having a mix of chiral forms that can include D-alanine, D-glutamic acid (D-glutamate), D-methionine, D-leucine, D-tyrosine, or D-phenylalanine (Science 325:1552, Sept. 18, 2009). (See the annotated bibliography, below, for other instances of D-amino acids found in various organisms.)
So does this fact solve the Chirality Problem for evolutionists, thus overthrowing a long-standing argument for creation and against a naturalistic chemical origin of life? The answer is no.
Although the debater’s statement had troubled me, it was some time before I conducted further research into right-handed amino acids in organisms. When I discovered the details in the technical literature, I realized that a naturalistic origin of life was just as problematic for evolutionists as it had ever been. Here’s what I learned:
(1) It remains true that only left-handed amino acids are built into proteins, the workhorses of all cells.
(2) Bacterial D-amino acids are actually produced from L-amino acids — by dedicated enzymes called “racemases.” Examples: D-alanine is formed from L-alanine by alanine racemase. D-glutamic acid can be formed either from L-glutamic acid (by glutamic acid racemase) or from a reaction involving D-alanine (by another enzyme, D-glutamate:D-alanine transaminase).
(3) These racemases (and other enzymes) themselves are proteins composed entirely of L-amino acids.
(4) D-amino acids are linked together by special enzymes called “ligases.” For example, two D-alanine molecules are often joined by D-alanine:D-alanine ligase.
(5) These ligase enzymes are proteins composed entirely of L-amino acids.
. . . In other words, D-amino acids are definitely second-class citizens! They do not come into bacterial cells on an equal basis with L-amino acids, nor does their presence increase the credibility of any evolutionary “primordial soup” scenario. Before there can be a D-amino acid there must be an L-amino acid as its precursor. D-amino acids completely depend on protein enzymes (made entirely of L-amino acids) for their origin and for their linkage to others of their kind. D-amino acids are not specified by the DNA code (as L-amino acids are), nor by the mRNA codons (as L-amino acids are), nor are they brought to a ribosome by specific tRNA molecules (as L-amino acids are) to be assembled into proteins.
The “Chirality Problem” therefore continues to be a frustrating unsolved enigma for evolutionists.

Spoof of hypothesized evolutionary “primordial soup” <http://www.mbio.ncsu.edu/jwb/PrimordialSoup.jpeg>
(Note the equal percentages of L- and D-amino acids, under “Nutrition Facts”.)
Annotated Bibliography
Articles highlighting the Chirality Problem:
Jon Cohen, “Getting All Turned Around Over The Origins of Life on Earth” (Science 267:1265f., Mar. 3, 1995).
[p. 1265] “The reason these molecules [DNA, RNA, amino acids] have such uniform handedness, or ‘chirality,’ is not known, but there is no shortage of theories on the subject. And, as was clear at a recent meeting on the topic in Los Angeles, there is also no shortage of passion, which is understandable, because the question of homochirality speaks to the mother of all scientific mysteries: the origin of life.
“Two dozen physicists, chemists, and astronomers journeyed from around the world to attend the 3-day [Feb. 15–17, 1995] meeting on ‘The Origin of Homochirality in Life,’ which several said was the first gathering devoted to the subject.”
Roger A Hegstrom and Dilip K. Kondepudi, “The Handedness of the Universe” (Scientific American 262(1):108-115, Jan. 1990).
[p. 109] “The catalytic ability of enzymes depends crucially on their three-dimensional structure, which in turn depends on their L-amino acid sequence. Synthetic chains of amino acids made of both L- and D-enantiomers do not twist in the way necessary for efficient catalytic activity; they cannot form the regular winding structure, called the alpha helix, that is present in most enzymes.” <http://quantummechanics.ucsd.edu/ph87/ScientificAmerican/Sciam/Hegstrom_The_Handedness_of_the_universe.pdf>
Articles describing attempts to solve the Chirality Problem:
Davide Castelvecchi, “Would You Eat Alien Pizza?” [original title: “Alien Pizza, Anyone?”] Science News 172(7):107, Aug. 18, 2007. <http://sciencewriter.org/2007/08/would-you-eat-alien-pizza/#more-88>
Explores several evolutionary proposals regarding the initiation of homochirality, and offers counterarguments to some of them. Ignores the fact that the natural tendency of any D/L mixture is always toward racemization and never toward amplification of one chiral form. (Amino acid racemization dating is based on this entropic tendency; see <http://en.wikipedia.org/wiki/Amino_acid_dating>. But see also creationist biochemist Duane Gish’s 1975 critique of this dating method: <http://www.icr.org/article/amino-acid-racemization-dating-method>.)
Jonathan Sarfati, “Origin of life: the chirality problem.” CEN Technical Journal [now titled TJ] 12(3):263-266, Dec. 1998. <http://creation.com/origin-of-life-the-chirality-problem>
A handy summary, and critique, of the various attempts to account for the origin of homochirality in living organisms — by a noted creationist who has an earned PhD in chemistry.
Alan Saghatelian et al., “A chiroselective peptide replicator.” Nature 409:797-801, Feb. 15, 2001. <http://www.ocf.berkeley.edu/~aathavan/libraire/pep-repl.pdf>
Before proposing their own solution to the chirality problem, the authors acknowledge: “The origin of homochirality in living systems is often attributed to the generation of enantiomeric differences in a pool of chiral prebiotic molecules, but none of the possible physiochemical processes considered [footnotes 1–7] can produce the significant imbalance required if homochiral biopolymers are to result from simple coupling of suitable precursor molecules.”
Then they write, “Here we report that a 32-residue peptide replicator, designed [N.B.!] according to our earlier principles, is capable of efficiently amplifying homochiral products from a racemic mixture of peptide fragments through a chiroselective autocatalytic cycle. . . . These results support the idea that self-replicating polypeptides could have played a key role in the origin of homochirality on Earth.”
Their “designed” 32-amino-acid replicator, however, was composed entirely of L-amino acids and would not have existed on an early Earth — nor would the specialized conditions they employed, such as strong solutions of guanidinium chloride (3M), sodium chloride (2M), and 3-(N-morpholino)-propanesulphonic acid (0.1M). And as famed evolutionary origin-of-life researcher Leslie Orgel remarked, “the substrates used in these experiments are complicated and could not have been prebiotic” (Rebecca Rawls, “Single-Handed Replication.” Chemical & Engineering News 79(8):15, Feb. 19, 2001).
Discussions of D-amino acids in organisms:
Kenneth Todar, “Structure and Function of Bacterial Cells: Cell Wall.” <http://www.textbookofbacteriology.net/structure_5.html>
A good standard introduction to bacterial cell walls, showing the incorporation of D-alanine and D-glutamate.
H. J. Rogers, H. R. Perkins, and J. B. Ward, Microbial Cell Walls and Membranes. London: Chapman and Hall, 1980. <http://www.springer.com/gp/book/9789401160162>
Includes detailed biochemical descriptions of the structure and biosynthesis of bacterial cell walls (pp. 190ff.). Recounts the involvement of D-amino acids and the enzymes that produce and link them.
Hubert Lam et al., “D-amino Acids Govern Stationary Phase Cell Wall Re-Modeling in Bacteria.” Science 325(5947):1552-1555, Sept. 18, 2009. <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2759711/pdf/nihms136087.pdf>
In addition to the D-amino acids commonly found in bacterial cell walls (D-alanine and D-glutamate), these researchers discovered others: D-methionine, D-leucine, D-tyrosine, and D-phenylalanine. All of these various D-amino acids are produced from the respective L-forms by racemase enzymes.
Yasuaki Saitoh et al., “Spatiotemporal Localization of D-Amino Acid Oxidase and D-Aspartate Oxidases during Development in Caenorhabditis elegans.” Molecular and Cellular Biology 32(10):1967-1983, 2012. <http://mcb.asm.org/content/32/10/1967.full.pdf>
The introduction contains a handy history of D-amino acid discoveries in a variety of organisms, including earthworms, silkworms, mammals, and lizards. Several functions and possible functions of these D-amino acids are mentioned. The authors suggest that the roundworm C. elegans can obtain some D-amino acids (D-alanine, D- aspartate, and D-glutamate) from its laboratory diet of E. coli bacteria, but it may use a serine racemase enzyme to manufacture D-serine from L-serine. (Such an enzyme has been found in yeast, and in some plants and mammals.) The extensive references section is a good source for further research on the topic of D-amino acids in various organisms.
Mendel Friedman, “Chemistry, Nutrition, and Microbiology of D-Amino Acids.” Journal of Agricultural and Food Chemistry 47(9):3457-3479, 1999. <http://pubs.acs.org/doi/pdfplus/10.1021/jf990080u>
Wide-ranging discussion of D-amino acids in various organisms, with a focus on the ones found in our food sources. A valuable supplement to the more technical article cited above.
